MAGNETITE EMBEDDED BIOCHAR AS NANO-SORBENT FOR EFFECTIVE ADSORPTION OF TEXTILE DYE
M. SEENUVASAN†, C.G. MALAR‡, R. CARTER§ and S. PRAVEEN*
†Department of Chemical Engineering, Hindusthan College of Engineering and Technology, Coimbatore, India.
Email: msvasan.chem@gmail.com; msvasan.chem@hicet.ac.in
‡ Department of Biotechnology, Rajalakshmi Engineering College, Thandalam, Chennai, India
Email: carlinmalar@gmail.com
§ Department of Electrical and Electronics Engineering, Rajalakshmi Engineering College, Thandalam, Chennai, India. Email: roncarterbus@gmail.com
* Department of Mechanical Engineering, Hindusthan College of Engineering and Technology, Coimbatore, India.
Email: tspraveenram@gmail.com
Cite this article as:
M. SEENUVASAN, C.G. MALAR, R. CARTER and S. PRAVEEN (2021) “MAGNETITE EMBEDDED BIOCHAR AS NANO-SORBENT FOR EFFECTIVE ADSORPTION OF TEXTILE DYE”, Latin American Applied Research, 51 (3), pp 185-192.
Abstract-- The nano-sorbent was synthesized by the embedment of magnetite onto the biochar obtained from Cassia auriculata for the effective adsorption of Levafix blue (LB) dye. Different instrumental techniques revealed the properties of biochar and the nano-sorbent. It was very distinct that the nano-sorbent gained highly favorable properties to be an effectual bio-sorbent. The effect of contact time, initial dye concentration and nano-sorbent dosage on the removal of LB dye was examined. Also, out of the kinetics studies models, the best fit and highest R2 values (0.9873) showed that the adsorption followed pseudo-second-order kinetics. Langmuir, Freundlich and Temkin isotherm models were established for the adsorption equilibrium data and the Temkin model showed the best reliability with the experimental results with highest R2 value of 0.9915. The adsorption system was modelled using the Artificial Neural Network (ANN) for biochar and nano-sorbent. The developed well-trained neural structure suggested the high performance of nano-sorbent.
Keywords-- Adsorption; Biochar; Artificial Neural Network; Kinetics; Isotherms.
I. INTRODUCTION
A major environmental pollution caused by hasty industrial enterprise and urbanization is the threat of aquatic life and human beings (Guler et al., 2019). This is mainly due to the fact that the contaminants are disposed into water bodies from various industries like paper and pulp, dye industry, tanneries, food processing, paint industry and cosmetics (Luo et al., 2011). Amongst the aforementioned industries, impact on the environment by the textile industries is higher. In general, waste water from textile industries are dispersible, hard-to-treat, high-volume, and hazardous (Jegatheesan et al., 2016). Also, the aquatic flora and fauna growth are highly inhibited by the reduced biological oxygen demand (BOD) in water caused by the effluent’s restriction of sunlight entry to water (Mayanglambam and Neelam, 2020). This is mainly because of the properties of dyes such as easy visibility to naked eye, stable molecules, toxic and non-biodegradable (Sivakumar et al., 2020).
Recently, synthetic dyes are most widely used for dyeing purposes in textiles (Waghmode et al., 2012; Kumar et al., 2017). The components present in these dyes are salts, surfactants, organic, inorganic compounds and polymers which are difficult to degrade (Kumar et al., 2017). Various conventional methods like coagulation- flocculation, ozonation, aerobic and anaerobic degradation, adsorption, membrane technology and electrochemical treatment (Tonucci et al., 2015; Vinodhini et al., 2015) are available for textile effluent treatment. Out of all these methods, adsorption is found to be an effective process for dye removal due to its low cost, ease of operation and sludge free process (Przystas et al., 2018).
Activated carbon from biochar is the most widely used adsorbent for dye removal due its porosity, high degree of selectivity and high surface area (Ferreira et al., 2020). Many studies were reported on preparation of low cost activated carbon from agricultural waste, lignocellulosic materials based on the carbon content of the material (Cepada et al., 2014). The physical activation through carbonization or chemical activation through chemical agents is usually performed. Though the adsorption of dye is at maximum rate by the activated carbon, regeneration becomes more tedious. In order to make the recovery of the adsorbent much easier, the researchers would gain interest towards the magnetic carriers (Malar et al., 2018).
Also, it is very important to evaluate the adsorption process statistically for efficient results. For an advanced and accurate prediction, a well-known statistical tool involving artificial intelligence (AI) like artificial neural networks (ANN) can be used. These AI techniques would decrease the number of experimental trials compared to that of conventional approaches (Tavakohlipour and Mokhtarian, 2014). Relatively, ANN has an outstanding ability in modelling because of its multi-training algorithms, higher speed and more precise prediction using complex relations (Tu, 1996).
Hence in the present study, low cost activated carbon was prepared from Cassia auriculata precursor by chemical activation method and it is embedded to magnetite nanoparticle through co precipitation. It has been effectively used for the removal of Levafix Blue (LB) dye from textile effluent. The characteristics of nano-sorbent was completely analyzed through various instrumental techniques. The adsorption studies were carried out by varying the parameters like composite dosage, contact time and dye concentration in batch mode. Adsorption kinetics and isotherm were performed to analyze the best fit of the obtained data. ANN was employed in modelling an integrated neural structure using MATLAB Simulink and NN tool in order to determine the effectiveness of the nano-sorbent.
II. METHODS
A. Chemicals
The pods of Cassia auriculata were collected from the local community. Ferric chloride (FeCl3) and Ferrous sulphate (Fe2SO4) were obtained from Loba Chemie Pvt. Ltd. (Mumbai, India). Sodium hydroxide (NaOH) and phosphoric acid were purchased from Merck (Mumbai, India). Highest pure and analytical grade chemicals were used for the study.
B. Preparation of Biochar
Cassia auriculata fruit pod was selected as a precursor for the preparation of biochar. The collected pods were dried in an oven at 423 K for 24 h and then crushed into powder to a particle size of 1 to 5 mm. Impregnation ratio of 1:1 was maintained for mixing the sample and phosphoric acid, the mixture was kept in atmospheric condition for 1 h. Then the process of carbonization was performed by placing the mixture in a muffle furnace at 773 K for 1 h. Carbonized material was washed with distilled water until its pH became constant. It was dried, stranded and stored in a container.
C. Embedment of magnetite onto the biochar
Magnetite was embedded onto the biochar through co-precipitation method. Briefly, FeCl3 and FeSO4 solution was mixed and agitated vigorously in a magnetic stirrer at 333 K for 30 min followed by the addition of biochar. The embedding process was favored by vigorous stirring for 40 min and addition of 2 M NaOH solution was added to the suspension for the precipitation process under continuous stirring. The solution was agitated for 1 h and maintained at atmospheric condition for 24 h after which, the solution was washed and dried in an oven.
D. Instrumental Analysis
The properties of the prepared biochar before and after embedding magnetite nanoparticles were studied using different instrumental analysis. The surface morphology of the nano-sorbent was analyzed through Field Emission Scanning Electron Microscope (FESEM, FEI quanta FEG 200, operating voltage 30 kV). The functional groups to confirm the biochar formation and magnetite embedment were performed using Fourier Transform Infrared Spectroscopy (FTIR) at wavenumbers between 400-4000 cm-1 (Perkin Elmer FTIRC 100566). The crystallite structure was analyzed through the X – ray Diffraction (XRD, X’Pert3 Powder X-Ray Diffractometer). The porous behavior of the nano-sorbent was understood from the Brunauer-Emmett-Teller (BET, Quantachrome ASiQwin) analysis. The embedment of magnetite nanoparticles onto the biochar was also confirmed through the magnetic property analysis using Vibrating Sample Magnetometer (VSM Lakeshore 7407, United States)
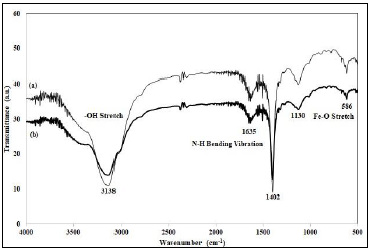
Figure 1: FTIR Spectrum of (a) Biochar and (b) Nano-sorbent.
E. Batch Adsorption Studies
The adsorption of the LB dye was studied as batch experiments using suspension of 0.5 g of nano-sorbent in various concentrations (10 – 60 mg L-1) of dye solution. The reaction was performed at room temperature with agitation at constant speed of 100 rpm for 60 min. The effect of contact time (5-60 min), dye concentration (10-60 mg L-1) and nano-sorbent dosage (0.5-2.5 g) were evaluated. At regular time intervals, residual concentration of dye in supernatant solution was analyzed by using UV Spectrophotometer at maximum wavelength of 613 nm. Equation (1) was used to determine the percentage removal of dye (Seenuvasan et al., 2018a).
![]() (1)
(1)
Where ![]() is the initial concentration of
LB dye in solution (mg L-1) and
is the initial concentration of
LB dye in solution (mg L-1) and ![]() is the
equilibrium concentration of LB dye in solution (mg L-1).
is the
equilibrium concentration of LB dye in solution (mg L-1).
III. RESULTS
A. FTIR Analysis
Figure 1 depicts the FTIR spectrum of biochar (a) and the magnetite embedded biochar nano-sorbent (b). The peak at 3138 cm-1 indicated the presence of the -OH group in both biochar and nano-sorbent. The peak at 1635 cm-1 showed the –NH bending vibrations and the shift at this peak in the nano-sorbent confirmed the embedment of magnetite (Malar et al., 2019a). The magnetite embedment on the biochar was also proved by the presence of a characteristic peak at 586 cm-1 corresponding to the Fe-O bond (Seenuvasan et al., 2018b).
B. XRD Analysis
The XRD patterns of biochar and nano-sorbent are shown in fig. 2 (a) and (b). The XRD patterns of biochar were observed with peaks at 2θ = 9.4 °, 14.0 ° and 16.1 °. The peaks at 36.12 °, 44.62 ° and 63.41 ° in the diffraction pattern of nano-sorbent indicated the magnetite coating on the biochar. Also the crystallite structure of the nano-sorbent was found to be a spinel structure from the characteristic peak with indices (3 1 1). The broader peak and the sharper peak showed the smaller size and crystallinity of the nano-sorbent (Balaji et al., 2016). The Debye-
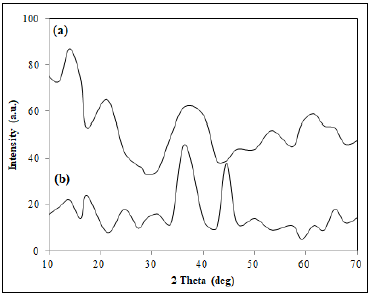
Figure 2: XRD pattern of (a) Biochar and (b) Nano-sorbent.

Figure 3: FESEM images of (a) Biochar and (b) Nano-sorbent
Scherrer equation (![]() ) was used to determine the average
size of the nano-sorbent and was found to be 24±0.65nm.
) was used to determine the average
size of the nano-sorbent and was found to be 24±0.65nm.
C. FESEM Analysis
Figure 3 (a) and (b) illustrates the FESEM image of biochar and nano-sorbent respectively. The image clearly showed the network-like porous morphology of the biochar.
Homogenous cross-linkage and the presence of pores on the surface of the biochar were observed. The electrograph of nano-sorbent showed its defined structure and embedment onto biochar with an average size of 29.42±0.31 nm. It was found to be in agreement with the size evaluated from XRD diffraction. Also the rough surface suitable for efficient adsorption was also observed in the FESEM image of nano-sorbent.
D. BET Analysis
The porous property of biochar and the nano-sorbent was detected from N2 adsorption/desorption isotherm. The specific surface areas were determined to be 95.85 m2/g and 85.04 m2/g for biochar and nano-sorbent respectively. Defined particle size were clear from the outward reduction in the surface area. Pore size distribution exhibited the broad porosity of nano-sorbent with pore size in 5-10 nm range. Also, higher value for constant C was observed for nano-sorbent (182.42) which confirmed its porous nature (Sing, 2001). Hence from the BET analysis, the surface porous nature of nano-sorbent which is beneficial for an effective adsorption was very evident.
E. VSM Analysis
Super paramagnetism of the nano-sorbent was observed from the magnetization hysteresis curve as depicted in Fig. 4. Lower coercivity and retentivity were found in the absence of magnetic field (Hwang et al., 2004). The incorporation of magnetic behavior to the biochar would be very effective in recovery of the sorbent after the process.
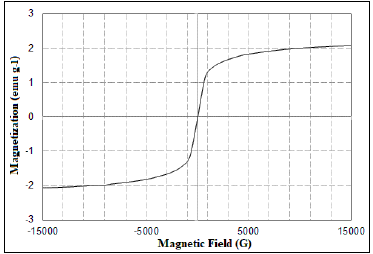
Figure 4: VSM Magnetization curve.
F. Analysis of Adsorption Kinetics
The adsorption of LB dye was found to be highly dependent on the nano-sorbent dosage and the contact time. Adsorption of the LB dye was observed to be rapid, followed by a steady decrease and equilibrium.
Nano-sorbent exhibited a longer time of 50 min to attain equilibrium, whereas the biochar reached equilibrium at 30 min signifying its lower adsorption capacity. Maximum adsorbed amount of 30 mg g-1 at equilibrium was observed for nano-sorbent than that of biochar (10 mg g-1). This indicated the higher affinity of LB dye towards nano-sorbent even with the reduced specific surface area.
The effect of contact time (mins), initial dye concentration (mg. L-1) and adsorbent dosage (g) on the % removal of dye is depicted in the Fig. 5 (a), (b) and (c) respectively. On comparison with the biochar, nano-sorbent was observed to perform the adsorption at better rate. With the embedment of magnetite nanoparticles, there was a significant increase in the adsorption capacity. This can be attributed to the fact that more adsorption site was resulted along with the surface alterations induced by magnetite (Simsek et al., 2015).
The possible kinetic models include first-order, pseudo-first-order, pseudo-second-order. With the relation between the experimental data and the predicted data, the correlation coefficient (R2) values were determined. The model with highest R2 value describes the kinetics of the successful adsorption of LB dye.
The Eqs. (2), (3) and (4) describe the expressions of first-order, pseudo-first-order and pseudo-second-order models respectively (Malar et al., 2019b).
![]() (2)
(2)
where ![]() and
and ![]() are the
concentration of LB dye (mg/L) at initial and at time
are the
concentration of LB dye (mg/L) at initial and at time ![]() respectively,
respectively, ![]() is
the first-order rate constant (min-1)
is
the first-order rate constant (min-1)
![]() (3)
(3)
Where ![]() and
and ![]() are the
concentration of LB dye at equilibrium and at time t respectively and
are the
concentration of LB dye at equilibrium and at time t respectively and ![]() is
the pseudo-first-order rate constant.
is
the pseudo-first-order rate constant.
.
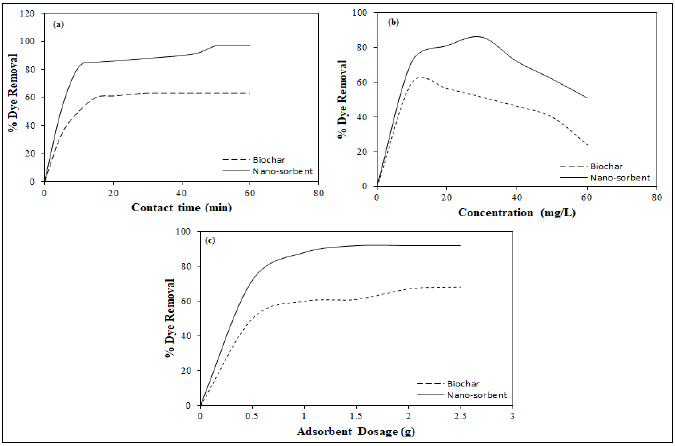
Figure 5: Effect of (a) Contact time (b) Initial dye concentration and (c) Biochar and nano-sorbent dosage.
![]() (4)
(4)
where ![]() is the rate constant for
pseudo-second-order kinetics.
is the rate constant for
pseudo-second-order kinetics.
The rate constants were determined from the slope and intercepts of the plots and are listed in Table 1. Figure 6 (a-c) illustrates the kinetic models of first-order, pseudo-first-order and pseudo-second-order respectively. Linear fit and highest R2 value of 0.9873 was obtained for pseudo-second-order model suggesting its applicability for the system. Hence it was confirmed that the adsorption process of LB dye on the nano-sorbent follows the pseudo-second-order kinetics. This showed that LB dye was adsorbed onto the nano-sorbent through chemisorption which involve the reaction on the surfaces of both the adsorbent and adsorbate.
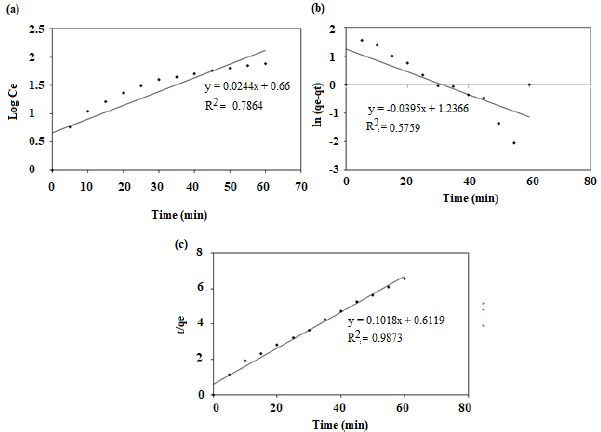
Figure 6: Kinetic models (a) First-order (b) Pseudo-first-order (c) Pseudo-second-order and their statistical parameters.
Table 1. Kinetic Models and their Statistical Parameters

G. Adsorption Isotherm
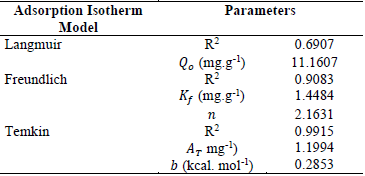
The equilibrium relationship between the adsorbent and adsorbate and their interactions are understood by the adsorption isotherm models. In order to describe the behavior of adsorption equilibrium data for the adsorption of LB dye onto nano-sorbent, three isotherm models such as Langmuir (Langmuir, 1998), Freundlich (Freundlich, 1906) and Temkin (Hamdaoui and Naffrechoux, 2007) were used.
Equation (5) describes the well-known two parameter Langmuir model
![]() (5)
(5)
where ![]() and
and ![]() are the
adsorption capacity (mg.g-1) at equilibrium and maximum
respectively,
are the
adsorption capacity (mg.g-1) at equilibrium and maximum
respectively, ![]() is the equilibrium concentration
(mg.L-1) and
is the equilibrium concentration
(mg.L-1) and ![]() is the Langmuir constant (L.mg-1).
is the Langmuir constant (L.mg-1).
Equation (6) describes the linear form of Freundlich model
![]() (6)
(6)
where ![]() (mg.g-1) and n are
the Freundlich constants that give the measure of adsorption capacity and
intensity respectively.
(mg.g-1) and n are
the Freundlich constants that give the measure of adsorption capacity and
intensity respectively.
Temkin isotherm that describes the adsorbent-adsorbate interactions can be expressed as given in Eq. (7)
Table 2. Parameters obtained from various adsorption isotherm models.
![]() (7)
(7)
where R is the universal gas constant (8.314
J.mol-1.K-1), T is the temperature at 298 K, b is the
constant related to heat of sorption (J.mol-1) and ![]() is
the Temkin constant (L.g-1). The plots of 1/qe Vs 1/Ce
(Fig. 7a),log qe Vs log Ce (Fig. 7b) and
is
the Temkin constant (L.g-1). The plots of 1/qe Vs 1/Ce
(Fig. 7a),log qe Vs log Ce (Fig. 7b) and ![]() Vs
ln
Vs
ln ![]() (Fig. 7c) are used to obtain the
parameters of Langmuir, Freundlich and Temkin isotherm models respectively and
are listed in Table 2.Temkin isotherm model described the data and found to
best fit with highest R2 value. This showed the occurrence of
multilayer adsorption of LB dye on the surface of nano-sorbent. The
heterogenous surface of adsorbent and the distribution of the sorption heat and
adsorbate were confirmed through the fit of Temkin isotherm model.
(Fig. 7c) are used to obtain the
parameters of Langmuir, Freundlich and Temkin isotherm models respectively and
are listed in Table 2.Temkin isotherm model described the data and found to
best fit with highest R2 value. This showed the occurrence of
multilayer adsorption of LB dye on the surface of nano-sorbent. The
heterogenous surface of adsorbent and the distribution of the sorption heat and
adsorbate were confirmed through the fit of Temkin isotherm model.
The nano-sorbent exhibits better performance in adsorbing LB dye with comparison to the similar researches (Bouyakoub et al., 2011; ELayazi et al., 2014). In addition, the magnetite embedment facilitates the easy recovery of the adsorbent using external magnetic field.
H. Modelling of Adsorption System Using ANN
Modelling of the adsorption system was carried out using a multilayer perceptron with the obtained data. The neural network was trained with the experimental data of ad-
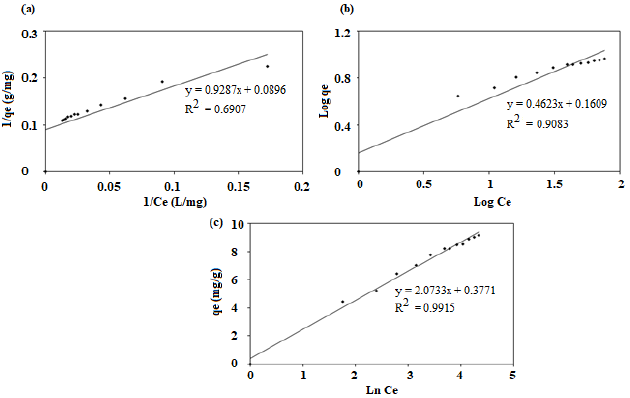
Figure 7: Equilibrium modeling (a) Langmuir isotherm (b) Freundlich isotherm (c) Temkin isotherm plots for adsorption of LB dye.
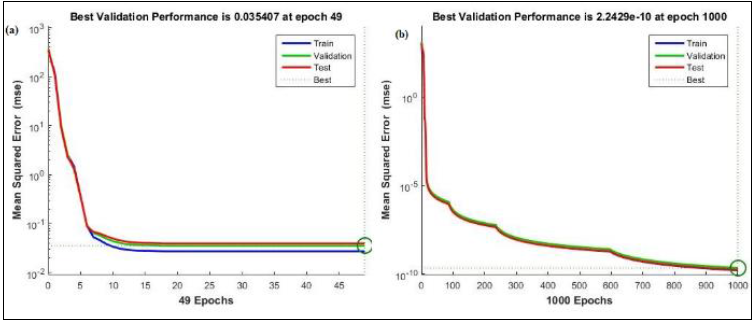
Figure 8: MSE of (a) Biochar and (b) Nano-sorbent
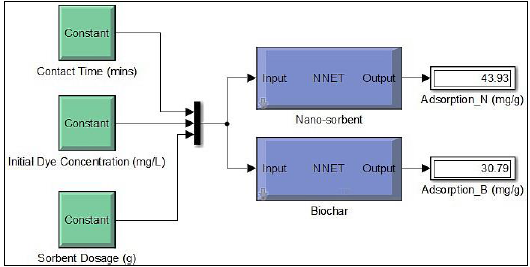
Figure 9: Structure of ANN modelled for the adsorption study
Table 3.
Experimental data used in training ANN.
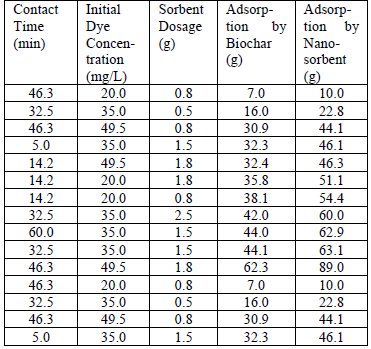
sorption and an effective neural structure was modelled for both biochar and nano-sorbent (Table 3). The predicted data of nano-sorbent was found to best fit the experimental data that was seen from the correlation coefficient value equal to 1. Also, maximum training was performed with the experimental data of biochar with correlation coefficient value much closer to 1.
Figure 8 shows the mean square error (MSE) of biochar and nano-sorbent. Very minimal error values of 0.035407 and 2.2429e-10 for biochar and nano-sorbent respectively. This showed that the modelled neural network would generate the output with least errors. The ANN model developed for the adsorption system by biochar and nano-sorbent with hidden layers and number of neurons. It shows the neural structure with three input parameters such as contact time (mins), initial dye concentration (mg/L) and sorbent dosage (g) with the adsorbent rate (mg/g) as the output as illustrated in Fig. 9. With the test data provided, the adsorption rate of nano-sorbent was observed to be higher than that of biochar which was found to be in agreement with the experimental data.
IV. CONCLUSIONS
Biochar was prepared from pods of Cassia auriculata and the magnetite was embedded onto it to synthesize the nano-sorbent. Various analytical techniques were employed to gain insight knowledge on the characteristics of biochar and nano-sorbent. The average size of the nano-sorbent was found to be 24±0.65nm from XRD and FESEM results. FT-IR peaks and VSM plot confirmed the embedment of magnetite onto biochar and the surface area was clearly evaluated from the BET results. Nano-sorbent exhibited higher contact time required to attain equilibrium which proved its efficiency. Pseudo-second-order kinetics was found to be the best fitting kinetics model which showed that the adsorption process has followed chemisorption. Amongst the isotherm models studied, Temkin isotherm was found to be the best fit with highest regression coefficient of 0.9915.The best performance in adsorption of LB dye by the nano-sorbent was also observed from the well-trained neural structure by ANN with least MSE.
REFERENCES
Balaji, N., Kumar, K.S., Seenuvasan, M., Vinodhini, G., and Kumar, M.A. (2016) Immobilization of laccase onto micro-emulsified magnetic nanoparticles for enhanced degradation of a textile recalcitrant. J. Environ. Biol. 37, 1489-1496.
Bouyakouba, A.Z., Lartiges, B.S., Ouhibb, R., Kacha, S, El Samrani, A.G., Ghanbaja, J., Barres, O. (2011). MnCl2 and MgCl2 for the removal of reactive dye Levafix Brilliant Blue EBRA from synthetic textile wastewaters: An adsorption/aggregation mechanism. J. Hazard. Mater. 187, 264-273.
Cepada, E.A., Iriarte, U., and Sierra, I. (2014) Kinetic and Thermodynamic Study of Chlorobenzene Adsorption from Aqueous Solutions onto Granular Activated Carbon. Lat. Am. Appl. Res. 44, 41-147.
ELayazi, L., Ellouzi, I., Khairat, A., EL Hajjaji, S., Mountacer, H. (2014) Removal of blue levafix dye from aqueous solution by clays. J. Mater. Environ. Sci. 5, 2030-2036.
Ferreira, A.B., Baierle, A.L., Zazycki, M.A., Perondi, D., Godinho, M., Foletto, E.L., Dotto, G.L., and Collazzo, G.C. (2020) Biochar Pecan Nutshell via Pyrolysis for Use as an Adsorbent: An Applied Study for the Removal of Hexavalent Chromium. Lat. Am. Appl. Res. 50, 203-208.
Freundlich, H.M. (1906) Over the adsorption in solution. J. Phys. Chem. 57, 385-470.
Guler, M., Cetintas, S., and Bingol, D (2019) Cinnamon bark as low-cost and eco-friendly adsorbent for the removal of indigo carmine and malachite green dyestuffs. Int. J. Environ. An. Ch. 101, 735-757.
Hamdaoui, O., and Naffrechoux, E. (2007) Modeling of adsorption isotherms of phenol and chlorophenols onto granular activated carbon Part II. Models with more than two parameters. J. Hazard. Mater. 147, 401-411.
Hwang, S., Lee, K., Park, J., Min, B., Haam, S., Ahn, I., and Jung, J. (2004) Stability analysis of Bacillus stearothermophilus L1 lipase immobilized on surface modified silica gels. Biochem. Eng. J. 17, 85-90.
Jegatheesan, V., Pramanik, B.K., Chen, J., Navaratna, D., Chang, C.Y., and Shu, L. (2016) Treatment of textile wastewater with membrane bioreactor: A critical review, Bioresour. Technol. 204, 202-212.
Kumar, M.A., Poonam, S., Kumar, V.V., Baskar, G., Seenuvasan, M., Anuradha, D., and Sivanesan, S. (2017) Mineralization of aromatic amines liberated during the degradation of a sulfonated textile colorant using Klebsiella pneumoniae strain AHM, Process Biochem. 57, 181-189.
Kumar, M.A., Priyadarshini, R., Nilavunesan, D., Seenuvasan, M., Kumar, V.V., Anuradha, D., Sivanesan, S. (2016) Biotransformation and detoxification of a greater tinctorial textile colorant using an isolated bacterial strain, J. Environ. Biol. 37, 1497-1506.
Langmuir, I. (1998) The adsorption of gases on plane surface of glasses. J. Am. Chem. Soc. 40, 1361-1403.
Luo, Y., Xu, L., Rysz, M., Wang, Y., Zhang, H., and Alvarez, P.J.J. (2011) Occurrence and Transport of Tetracycline, Sulfonamide, Quinolone, and Macrolide Antibiotics in the Haihe River Basin, China, Environ. Sci. Technol. 45, 1827-1833.
Malar, C.G., Seenuvasan, M. and Kumar, K.S. (2018) Prominent Study on Surface Properties and Diffusion Coefficient of Urease-Conjugated Magnetite Nanoparticles. Appl. Biochem. Biotechnol. 186, 174-185.
Malar, C.G., Seenuvasan, M. and Kumar, K.S. (2019a) Adsorption of nickel ions by surface modified magnetite nanoparticles: Kinetics study. J. Environ. Biol. 40, 731-735.
Malar, C.G., Seenuvasan, M. and Kumar, K.S. (2019b) Improvisation of diffusion coefficient in surface modified magnetite nanoparticles: A novel perspective. Mater. Sci. Engg. C. 103, 109832.
Mayanglambam, B. and Neelam, S.S. (2020) Physicochemistry and water quality of Loktak Lake water, Manipur, India. Int. J. Environ. An. Ch. DOI: 10.1080/03067319.2020.1742888.
Przystas, W., Zablocka-Godlewska, E., and Grabinska-Sota, E. (2018) Efficiency of Decolorization of different dyes using fungal biomass immobilized on different solid supports, Braz. J. Microbiol. 49, 285-295.
Seenuvasan, M., Suganthi, J.R.G., Sarojini, G., Malar, G.C.G., Priya, M.E., and Kumar, M.A. (2018a) Effective utilization of crustacean shells for preparing chitosan composite beads: Applications in ameliorating the biosorption of an endocrine disrupting heavy metal. Desal. Wat. Treat. 121, 22-27.
Seenuvasan, M., Vinodhini, G., Malar, C.G., Balaji, N and Kumar, K.S. (2018b) Magnetic nanoparticles: a versatile carrier for enzymes in bio-processing sectors. IET Nanobiotechnol. 12, 535-548.
Simsek, E.B., Kaya, I.G.B., and Beker, U. (2015) Comparison of Cr(VI) Adsorption onto Polymer-Based Magnetic Hybrid Adsorbents by Nonlinear Isotherm and Kinetic Models. Lat. Am. Appl. Res. 45, 27-32.
Sing, K. (2000) The use of nitrogen adsorption for the characterisation of porous materials. Colloids Surf. A Physicochem. Eng. Asp. 187-188, 3-9.
Sivakumar, D., Parthiban, R., Kumar, P.S. and Saravanan, A. (2020) Synthesis and characterization of ultrasonic-assisted Delonix regia seeds: modelling and application in dye adsorption, Des. Water Treat. 173, 427-441.
Tavakohlipour, H., Mokhtarian. (2014) Application of Neural Network for Estimation of Pistachio Powder Sorption Isotherms. Lat. Am. Appl. Res. 44, 189-194.
Tonucci, M.C., Gurgel, L.V.A., and de Aquino, S.F. (2015) Activated carbons from agricultural byproducts (pine tree and coconut shell), coal, and carbon nanotubes as adsorbents for removal of sulfamethoxazole from spiked aqueous solutions: Kinetic and thermodynamic studies, Indus. Crops Products. 74,
111-121.
Tu, J.V., (1996) Advantages and disadvantages of using artificial neural networks versus logistic regression for predicting medical outcomes. J. Clin. Epidemiol. 49, 1225-1231.
Vinodhini, G., Kumar, M.A., Balamanikandan, S., and Seenuvasan, M. (2015) Assessment of antimicrobial property of a secondary metabolite produced by an enriched bacterial culture isolated from soil, Res. J. Pharm. Tech. 8, 51-53.
Waghmode, T.R., Kurade, M.B., Kagalkar, A.N., and Govindwar, S.P. (2012) Differential fate of metabolism of a disperse dye by microorganisms Galactomyces geotrichum and Brevibacillus laterosporus and their consortium GG-BL, J. Environ. Sci.-China. 24, 1295-1304.
Received: September 26, 2020
Sent to Subject Editor: January 12, 2021
Accepted: May 16, 2021
Recommended by Subject Editor José Luis Díaz de Tuesta