Abstract-- This work
study the application of Taguchi method and factorial design of experiment for
modeling and optimization of the influence of some variables in the extraction
of essential oil of cloves by a lab scale microwave. This methodology was
compared with conventional methods such as hydrodistillation, steam
distillation, continuous solid-liquid extraction. Subsequently, the eugenol
present in clove oil was isolated, purified and characterized by gas
chromatography. Results showed that the yields obtained by microwave are
highest respect to others methods being the optimum conditions to produce a
92.4 % of yield of eugenol a temperature of 70 °C, with a stirring of 480 rpm,
in 25 minutes of process, using ethanol as solvent and a relation liquid-solid
of 19 mL/mg.
Keywords-- Taguchi, Factorial Design, ANOVA
Microwave-Assisted Extraction, Eugenol.
I. INTRODUCTION
Essential oils are aromatic
and volatile oil liquids extracted from different parts of plants (Guntero et al., 2018), which usually
consist of terpenes, terpenoids, alcohols, aldehydes, esters, ethers, ketones,
phenols (Ali et al., 2015). They are used in
aromatherapy, preservation of foods and as antimicrobial, analgesic, sedative,
anti-inflammatory, spasmolytic and local anesthesia (Bakkali et al., 2008; Amorati et al., 2013). Particularly,
many phenylpropenes impart a characteristic aroma, and some fresh fruits and
processed products that contain them are widely consumed (Koeduka et al., 2013). Phenylpropanoids
are a group of plant secondary metabolites and perform a vital role in plant communication,
pollinator attraction and defense against herbivores and other pathogens (Anand et al., 2016). Eugenol, a
phenylpropene is found in bay leaves, allspice, and the oil of cloves that
originate from the Syzygium species (Ito et al., 2005). Has wide
application in pharmaceuticals, cosmetics, dentistry, food, agriculture,
pesticides (Shahavi et al., 2015) and like a component
of the packaging of aliment or cosmetic with antioxidant properties (Guntero et al., 2019).
Clove
(Syzygium aromaticum; synonym: Eugenia cariophyllata) is an
important aromatic spice. Its essential oil contains mainly eugenol, and
smaller amounts of eugenyl acetate and the terpenoid β- caryophyllene organic
compounds (Ágilar-González and López-Malo, 2013). The
traditional methods for the isolation and purification of chemical constituents
from plants tissues require relatively long extraction time and big solvent
amounts. Moreover, many natural products are thermally unstable and may degrade
during thermal extraction and/or distillation (Kimbaris et al., 2006), which
requires knowing the best extraction conditions. In fact, process engineers
want to determine the levels of the design parameters at which the response
reaches its optimum. The optimum could be either a maximum or a minimum of a
function of the design parameters (Aslan, 2007). On the other hand, is known
that the microwave-assisted extraction (MAE) is one of the best green technologies
with advantages like, high extraction efficiency, good reproducibility,
low consumption of organic solvents and time and low carbon dioxide output. MAE
is based upon the selective and rapid localized heating of moisture in the
sample by microwaves. Due to the localized heating, pressure builds up within
the cells of the sample, leading to a fast transfer of the compounds from the
cells into the extracting solvent (Mandal and Mandal, 2010; Sides et al., 2000). The matrix to be
extracted (usually water-rich) is mixed with a solvent having a low dielectric
constant, so that most of the heating effect will be
concentrated on the plant material (Cravotto et al., 2008).
Various
extraction parameters that have influence upon the extraction process
will depend on a number of steps controlling the transport of analytes from the
matrix to the bulk fluid. Generally these parameters include extraction
solvent composition, solvent volume, extraction temperature, extraction time,
system pressures and the matrix characteristics (including pH, water content
and total organic matter content) (Wang et al., 2007).
In
this research we study the technology of microwave-assisted extraction and
three conventional extraction methods: steam distillation (SD),
hydrodistillation (HD) and solid-liquid extraction (SLE) In the application of SLE was analyzed the time and solvents, in
the case of SD and HD the time was evaluated, while an experimental orthogonal
design of five factors (temperature, stirring, time, ethanol solution, liquid-solid
relation) was used to investigate the effects of parameters of
microwave-assisted extraction. The novel of this work was to optimize the
eugenol production by MAE. In this sense, Taguchi method was chosen because
reduce the number of experiments like that amount of chemicals and time and a standard orthogonal array L-25 was used to examine
a five factors system at five levels. Based on experimental parameters the
operating conditions that ensure the optimal recovery of eugenol were
evaluated.
II. METHODS
A. Plant Material
The buds of clove (Eugenia
caryophyllata) were obtained from the same batch of a local producer in the
province of Córdoba, Argentina. Prior to extractions, buds of clove were dried
at 40 ºC in stove until constant weight, ground in a mortar and stored in
plastic bags at room temperature, protected from light. The moisture content of
dried clove buds was 14.0±0.5%.
B.
Extraction Methods
Hydrodistillation
(HD)
Clove (30 g) was
submitted to HD with a Clevenger-type apparatus. The studied parameter was extraction
time. The essential oil/water mixture was collected in an Erlenmeyer and then
was subjected to extraction with dichloromethane in order to pass the eugenol
to the organic phase. Sodium hydroxide 5% was added to the organic phase so the
salt of eugenol was solubilized in water. The last phase was washed with
dichloromethane to eliminate the organic component that could be remaining.
Hydrochloric acid 10% was added to the aqueous phase to reconstitute a phenol,
and dichloromethane was added to extract it. Later, sodium sulfate was added to
the organic phase, the mixture was filter. Solvent was removed by
roto-evaporator. As a result, pure eugenol was obtained.
Steam Distillation (SD)
A simple laboratory
apparatus that consists of a steam generator, a 500 mL round-bottom flask, a
collected vessel, a refrigerant and a tube Florentine, was used to perform the
SD. Water was heated to produce steam which flowed through the round-bottom
flask which contained the sample (30 g). The mixture of essential oil and water
was condensed as it passed through the refrigerant and separated in the tube
Florentine. The studied parameter was extraction time. Eugenol was obtained to
the same methodology described in HD.
Continuous Solid-Liquid Extraction (SLE)
In this case, two
equipment were used, Soxhlet and Twisselman extractors. To select the optimal
solvent to extract the compounds of interest, initially, the experiments were
carried out with solvents of different polarities during the same period of
time. Once the optimal solvent was identified, the effect of extraction time
was analyzed.
The
sample (30 g) was transferred into a filter paper and inserted into to
the corresponding equipment. The plant material was extracted to different time
with 150 mL of selected solvent (ethanol). Vapors generated were condensed upon
contact with the refrigerant, dripping through the sample into the boiling solvent.
Once the extraction has been reached, solvent was recovered. Eugenol was
obtained to the same methodology described in HD.
Microwave-Assisted Extraction (MAE)
Sample extractions
were carried out in an Anton Paar Monowave 300. For each experimental run, 1 g
of cloves powder was used. Extractions were performed under different
extraction conditions, modifying the range of temperature variation (30 to 70°C),
the range of stirring (240 to 1200 rpm), the range of time (5 to 25 min), the
range of solution of ethanol (concentrations greater than 20%), and the range
of liquid-solid ratio (7 to 19 mL/mg). The established values were chosen under
the following criteria: ethanol is a good absorber of microwave radiation and
clove oil is soluble in this, the liquid-solid ratio is important because an
optimum ratio ensures homogeneous and effective heating, the chosen range was
based on the capacity of the vial. Excessive solvent causes poor microwave
heating as the microwave radiation would be absorbed by the solvent and
additional power is required. Low ratio of solvent in solid promotes mass
transfer barrier as the distribution of active compounds is concentrated in
certain regions which limits the movement of the compounds out of cell matrix (Zhang et al., 2011). After
completion of the extraction, vials were allowed to cool at room temperature.
The products were closed tightly and were stored at 8°C prior to further
analyses. Eugenol was obtained to the same methodology described in HD.
C.
Characterization
The concentrates
were analyzed by a Perkin Elmer gas chromatograph (GC) equipped with an
on-column injector, a flame ionization detector (FID), an PE-5% capillary
column (DF: 0.53 µm, ID: 0.55 µm, L: 30 m), using nitrogen as carrier gas, to
determinate the percent total peak-area of aroma compounds. Oven temperature
was programmed to 80 °C for 2 min, then programmed heating from 80 °C to 230 °C
at a rate of 6 °C/min, and at 230 °C for 2 minutes. Injector and detector
temperatures were 230 °C. The carrier gas was adjusted to a linear velocity of
24 mL/min. Methyl salicylate was used as internal standard (Wenqiang et al., 2007).
GC-MS
analysis were performed on a gas chromatograph Shimadzu GC-QP5000 mass
spectrometer equipped with a standard non-polar capillary column (DF: 0.25 µm,
ID: 0.25 mm, L: 30 m) in chromatographic conditions similar to GC. The carrier
gas, helium, was adjusted to a linear velocity of 1 mL/min. The identification
of compounds from the extracts was made based on fragmentation patterns
together with matching of the mass spectra which obtain from each sample and
compared with those in the NIST Mass Spectral Library (Katata-Seru et al., 2017).
D. Yield calculations
From the results obtained of extractions and subsequent
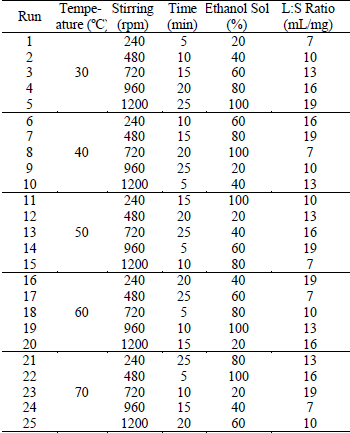
Table 1. Orthogonal design L25
[Sol = solution; L:S = Liquid:Solid]
analysis of GC, the
yield of essential oil (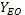 ) was calculated
by Eq. 1, yield of eugenol (YE) was calculated by Eq. 2, and yield of global process (YP) by Eq. 3 for each of the method applied. The values were expressed
as mean±standard deviation (n=2).
) was calculated
by Eq. 1, yield of eugenol (YE) was calculated by Eq. 2, and yield of global process (YP) by Eq. 3 for each of the method applied. The values were expressed
as mean±standard deviation (n=2).
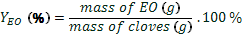 (1)
(1)
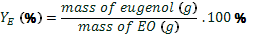 (2)
(2)
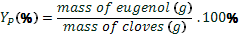 (3)
(3)
E. Experimental
design and statistical analysis
Experiments of MAE
were performed according to an experimental design of Taguchi of five levels
and five factors (L25) as shown in Table 1. This methodology provides a
complete knowledge of each factor and its effect on yields. Taguchi technique
analyzes the results by utilizing a statistical indicator referred as the
signal-to-noise ratio, (S/N ratio) that translates experimental information
into single value which shows presence of disparity (Kohli et al., 2018). The analysis of
variance (ANOVA) was used to investigate which factors had a remarkable result
on the response parameters and given optimum working conditions.
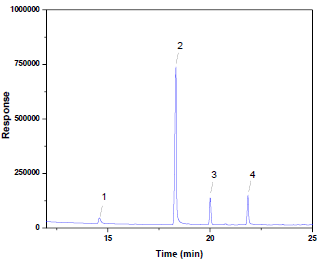
Figure 1: GC chromatogram obtained from analysis of oil
clove. [(1) methyl salicylate (the internal standard), (2) eugenol, (3)
β-caryophyllene and (4) eugenol acetate].
III. RESULTS
A. Chemical analysis of samples
Considering
theoretical data on the content of essential oils in vegetable species, up to
18 w% of essential oil could be obtained from clove flowers, being its main
components eugenol (89%), eugenol acetate and β-caryophyllene (5-15%)
(Cortés-Rojas, Fernandez de Souza and Oliveira, 2014). Although the composition
of the main components matches with data reported in the bibliography, such
high experimental values in the recovery of essential oil have not been
reported.
From
GC/GS-MS analysis of clove essential oil samples obtained by means of
extraction processes, three compounds have been identified: eugenol,
β-caryophyllene and eugenol acetate. Taking into account a relative
abundance of these compounds, it was found that eugenol is the main component.
A typical chromatogram of essential oils analysis is shown in Figure 1. Under
optimal MAE conditions, 80-92% of clove essential oil is eugenol, with 4-10%
β-caryophyllene and 8-14% eugenol acetate; even though lower
concentrations of these compounds have been determined in all of the samples,
maintaining the proportion between them. The results found in our studies are
within the range of concentrations and proportions of the components reported
by other authors in the analysis of clove oils (Jirovetz et al., 2006; Guan
et al., 2007).
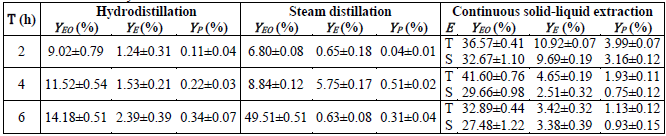
B. Conventional Extraction Methods
Conventional methods present limitations when analyzing the effect
of variables that can affect the process of
extracting essential
oil from a matrix. In the three unconventional methods evaluated in this work,
i.e. hydrodistillation, steam distillation and solid-liquid extraction, the variables
of temperature, matrix mass and solvent volume are both conditioned by the
properties of the stripping-extraction solvents used, and by the dimensions of
the equipment in which they are used.
Under
these conditions, the only variable to be evaluated is the extraction time,
which was analyzed at three levels: 2, 4 and 6 h. Trials were carried out at extraction
times greater than 6 h, whose results did not show higher yields than those
obtained at 6 h of the process. This could indicate that the extraction times
greater than 6 h would not increase the recovery of the essential oil and
therefore the operational yields.
Table
2 shows the essential oil, eugenol and process yields obtained by these
methods, at the three levels of the times selected. It should be noted that
both in HD and SD, it is water that is used as a solvent and therefore, the
process temperature is limited to its boiling point.
As can be seen for HD, the maximum EO, E and process
yields were obtained at 6 h of extraction. It can be observed that the EO
recovery yield increased 28% in 4 h and 57% in 6 h in relation to the yields
obtained at 2 h. In turn, it is observed that the EO extraction process was
accompanied by the enrichment of its content in eugenol, increasing the yield
to eugenol in 6 h within values of the order of 93% in relation to the values
obtained after 2 h of extraction. As a result of the process, the YP showed an increase of higher than 200% at 6 h of extraction, in
relation to the values found at 2 h.
In the case of SD, the maximum EO yields were obtained
at 6 h, while the maximum eugenol and process yields were obtained at 4 h. With
the increase in the extraction time, the EO yield raised. However, the process
was not accompanied by an enrichment of eugenol. This could indicate that,
under the extraction conditions throughout the process, the eugenol could have
been degraded by oxidation and/or polymerization of eugenol (Kapadiya et al.,
2018).
In the case of SLE, initially, the experiments were
carried out with solvents of different polarity at a same time (ethanol, water,
n-hexane), in order to compare the yields. The higher yield of eugenol was
obtained with ethanol. Once the solvent was selected, the effect of the time in
extraction process was evaluated with two different equipment: Twisselman
Extractor (T) an Soxhlet Extractor (S). As it is showed in Table 2, the results
indicate that regardless the type of extractor used, the results were similar.
Between 2 to 6 h of extraction, the EO yields were between 27-42%. Within just
2 h of extraction, the best YE and YP were obtained. Longer extraction times show a reduction in yields
of eugenol and process, which would indicate degradation of eugenol by
oxidation and polymerization during extraction times longer than 2 h (Kapadiya et
al., 2018).
From
the results presented in Table 2, it is possible to conclude that under the
conventional extraction methods studied, YP of 3.0-4.0% are obtained
by continuous S-L extraction at a process time of 2 h. The essential oils
obtained by this method presented higher eugenol contents, even those recovered
by the HD and SD procedures at different time tests.
C.
Microwave-Assisted Extraction and statistical analysis
In the Microwave
Assisted Extraction (MAE) technique there are several variables that influence
the process of extracting essential oil from cloves. For our case under study,
the factors evaluated were: temperature, stirring speed, extraction time,
extraction solvent and matrix mass/solvent volume ratio (liquid: solid ratio).
According to Taguchi, the design of the experiments was carried out including
these 5 factors at 5 levels. The selected levels for each factor were considered
on the basis of previous experiences of the working group.
On
the basis of the results obtained from the Taguchi experiment design, the mean
square standard deviation (MSD) and the signal-to-noise ratio (S/N) were calculated.
The S/N ratio is a performance measure developed by Taguchi to choose the best
levels as to deal with noise. In other words, the S/R ratio is a robustness
measure used to identify control factors that reduce the variability of the
extraction process by minimizing the effects of factors that cannot be
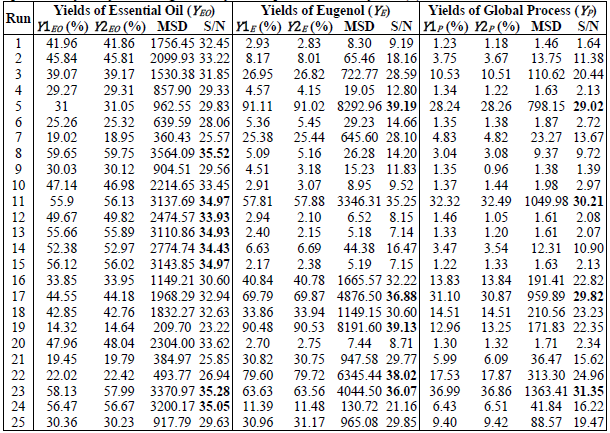
controlled (noise factors). Table 3 shows the results of the yields, MSD and
S/N ratio, for each experimental design conditions.
By
analyzing the results of the S/N ratio for the YEO,
it is observed that the maximum values are between 33.93-35.52, which
correspond to tests 8, 11, 12, 13, 14, 15, 23 and 24 (marked in bold), which
correspond to the best EO performance obtained with values between 49.7-56.7.
Most of the best results are observed between tests 11-15, which are carried
out at 50°C. A better analysis of the results is obtained by analyzing the
total values of the S/N ratio for each level of the evaluated factors, which is
presented in Table 4. The highest values of the S/N ratio indicate that optimal
conditions for the best YEO performance is: 50°C, 1200 rpm, 15 min, 40% ethanol solution and 7
mL/mg of S: L ratio.
At
the same time, the experimental yields of eugenol were analyzed and the results
are presented in Table 3. From the S/N ratio, it is observed that the maximum
values are in the range of 36.07-39.19, with YE in
the range of 63.56-91.11, whose conditions would correspond to that of tests 5,
17, 19, 22 and 23. Only test 23 equates with the best results obtained with YEO. For a better analysis of the results, we examined the total values
of the S/N relations for each level of the different factors in Table 4. The
results indicate that the optimal conditions to obtain maximum yields at E are:
70 ° C, 480 rpm, 25 min, 100% ethanol solution and 19 mL/ mg liquid-solid ratio.
The results that corresponds to the process yields are
shown in Table 3 and Table 4. Based on the analysis of the S/N relationships,
the best process yields correspond to tests 5, 11, 17 and 23. In these cases
the ranges of S/N and YP are between 29.02-31.25 and 28.24-36.99%, respectively. Only the
conditions of tests 17 and 23 matches the best performances previously
analyzed. Based on the analysis of the total values of the S/N ratios for each
level of the different factors, the optimal conditions to obtain the best
performance of YP are:
Table 4. Total S/N ratios corresponding to
results of yields
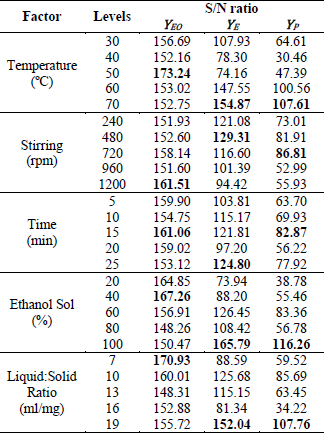
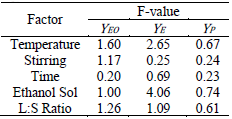
Table 5. Statistical F of experimental data
obtained for Taguchi design.
70°C, 720 rpm, 15
min, 100% ethanol solution and 19 mL/mg liquid-solid ratio. Similar levels of
the factors proved to be optimal when analyzing YEO
and YE.
In
order to analyze which of the factors under studies have a greater influence on
the extraction process, we evaluate the values of the F statistic. This test
allows us to assess the effect of the experimental factors on the average
response, compared to the experimental error. A high value of F associated with
a factor indicates that such a factor has a greater effect on performance, in relation
to the other factors in question; even if a low value of the F associated with
a factor, indicates a greater weight of such a factor on the results.
Table 5 shows the values of statistic F obtained with
different factors in relation to the experimental results of EO, E and process
yields. For YEO and YP, the lowest F value corresponds to that of the factor of time and
indicates that this variable is the least influential on the process
performance. In the case of the eugenol yield, the lower value of F corresponds
to stirring. For the whole of the returns of EO and E, the temperature factor
is predicted to be the most influential.
From the results analyzed it is possible to conclude
that high EO, E and process yields would be obtained
Table 6. Best yields of eugenol obtained by
different methods

under conditions
established by experience 23, although the factors can be properly selected on
the basis of the established process requirements.
It
is of interest to obtain essential oil of clove with high concentrations of
eugenol, it is for this reason that we consider the optimal conditions
established by the Taguchi design to obtain maximum YE.
Tests were carried out under these conditions, finding YE of 92.43±0.85.
C. Comparisons between yields obtain by different
methods
The selection of an extraction method and its parameters will depend
on the objective of production, costs, safety and environmental impact. As we
said previously, it is a requirement for our work to obtain clove essential oil
with high concentrations of eugenol. Table 6 shows the best YE results found by the extraction methods evaluated in this work,
including the experimental conditions in which they were obtained. From these
results it can be observed that the lowest YE
were obtained by the HD and SD processes, and the MAE process substantially exceeded
these yields. MAE is a method that offers huge advantages over conventional
methods, mainly the reduction in the times of extraction and the volume of
solvent extraction. Although the best eugenol yields were a consequence of high
YEO for the MAE experiments, this was not reflected on the tests
carried out through unconventional methods. The extracts obtained by using
non-conventional methods were blackish brown due to the presence of oxidation
and polymerization products as a consequence of thermal degradation;
characteristics that were not presents in the essential oils obtained by MAE.
IV. CONCLUSIONS
This investigation
is focused in the study of different parameters of extraction techniques to
obtain cloves oil and eugenol through purification of EO. The experimental
design of Taguchi allows optimizing the parameters of the MAE process. The
optimized conditions for obtaining of eugenol based on the five factors were 70
°C of temperature, with 480 rpm of stirring, 25 min of process, 100 % of
solvent and a liquid-solid ratio of 19 mL/mg. That means, MAE offers a
production of eugenol with lower energy cost and solvent, making it an
environmentally friendly technique.
AcknowledgEments
Authors thank the
Agencia Nacional de Ciencia y Tecnología (ANCyT) of Argentina, PICT 2014 No
1587 and by CAI+D 2017 PE N° 50420150100056LI of the Universidad Nacional del
Litoral, Santa Fe, Argentina for financial support of this work.
REFERENCES
Águilar-González, A.E. and López-Malo, A. (2013) Extractos y aceite
esencial del clavo de olor (Syzygium aromaticum) y su potencial aplicación como
agentes antimicrobianos en alimentos. Temas Selectos de Ingeniería de los
Alimentos. 7, 35–41.
Ali, B., Al-Wabel, N.A., Sham, S., Ahamad, A., Khan, S.A. and Anwar,
F. (2015) Essential oils used
in aromatherapy: A systemic review. Asian Pacific Journal of Tropical
Biomedicine. 5, 1–11.
Amorati, R., Foti, M.C. and Valgimigli, L. (2013) Antioxidant
Activity of Essential Oils. Journal of Agricultural and Food Chemistry. 61,
10835–10847.
Anand, A., Jayaramaiah, R.H., Beedkar, S.D., Singh, P.A., Joshi,
R.S., Mulani, F.A., Dholakia, B.B., Punekar, S.A., Gade, W.N., Thulasiram, H.V.
and Giri, A.P. (2016) Comparative functional characterization of eugenol
synthase from four different
Ocimum species: Implications on eugenol accumulation. Biochimica et Biophysica Acta. 1864, 1539–1547.
Aslan, N. (2007) Application of response surface methodology and
central composite rotatable design for modeling the influence of some operating
variables of a Multi-Gravity Separator for coal cleaning. Fuel. 86,
769–776.
Bakkali, F., Averbecka, S., Averbecka, D. and Idaomaret, M. (2008) Biological
effects of essential oils – A review. Food and Chemical Toxicology. 46,
446–475.
Cravotto, G., Boffa, L., Mantegna, S., Perego, P., Avogadro, M. and Cintas,
P. (2008) Improved extraction of vegetable oils under high-intensity ultrasound
and/or microwaves. Ultrasonics Sonochemistry. 15, 898–902.
Guan, W., Li, S., Yan, R., Tang, S. and Quan, C. (2007) Comparison
of essential oils of clove buds extracted with supercritical carbon dioxide and
other three traditional extraction methods. Food Chemistry. 101.
1558–1564.
Guntero, V.A., Ferretti, C.A., Mancini, P.M.E. and Kneeteman, M.N. (2018)
Synthesis and Encapsulation of bis-eugenol in a Mesoporous Solid Material:
Enhancement of the Antioxidant Activity of a Natural Compound from Clove Oil. Chemical
Science International Journal. 22, 1–10.
Guntero, V.A., Ferretti, C.A., Mancini, P.M.E. and Kneeteman, M.N. (2019)
Mesoporous Materials as Supports of Active Molecules: A Versatile Strategy Of
Functionalization of Inorganic Oxides. Biomedical Journal of Scientific
& Technical Research. 15, 8–11.
Ito, M., Murakami, K. and Yoshino, M. (2005) Antioxidant action of
eugenol compounds: role of metal ion in the inhibition of lipid peroxidation. Food
and Chemical Toxicology. 43, 461–466.
Jirovetz, L., Buchbauer, G., Stoilova, I., Stoyanova, A., Krastanov,
A. and Schmidt, E. (2006) Chemical composition and antioxidant properties of
clove leaf essential oil. Journal of Agricultural and Food Chemistry. 54,
6303-6307.
Kapadiya, S., Parikh, J., Desai, M. (2018) A greener approach
towards isolating clove oil from buds of Syzygium aromaticum using microwave
radiation. Industrial Crops & Products, 112, 626-632.
Katata-Seru, L., Lebepe, T.C., Aremu, O.S. and Bahadur, I. (2017) Application
of Taguchi method to optimize garlic essential oil nanoemulsions. Journal of
Molecular Liquids. 244, 279–284.
Kimbaris, A.C., Siatis, N.G., Daferera, D.J., Tarantilis, P.A.,
Pappas, C.S. and Polissiou, M.G. (2006) Comparison of distillation and
ultrasound-assisted extraction methods for the isolation of sensitive aroma
compounds from garlic (Allium sativum). Ultrasonics Sonochemistry. 13,
54–60.
Koeduka, T., Suzuki, S., Iijima, Y., Ohnishi, T., Suzuki, H., Watanabe,
B., Shibata, D., Umezawa, T., Pichersky, E. and Hiratake, J. (2013) Enhancement
of production of eugenol and its glycosides in transgenic aspen plants via
genetic
engineering. Biochemical and Biophysical Research Communications.
436, 73–78.
Kohli, H.P., Gupta, S. and Chakraborty, M. (2018) Extraction of
Ethylparaben by emulsion liquid membrane: Statistical analysis of operating
parameters. Colloids and Surfaces A. 539, 371–381.
Mandal, V. and Mandal, S.C. (2010) Design and performance evaluation
of a microwave based low carbon yielding extraction technique for naturally
occurring bioactive triterpenoid: Oleanolic acid. Biochemical Engineering
Journal. 50, 63–70.
Shahavi, M.H., Hosseini, M., Jahanshahi, M., Meyer, R.L. and Darzi,
G.N. (2015) Evaluation of critical parameters for preparation of stable clove
oil nanoemulsion. Arabian Journal of Chemistry. 12, 3225-3230.
Sides, A., Robards, K. and Helliwell, S. (2000) Developments in
extraction techniques and their application to analysis of volatiles in foods. 19,
322–329.
Wang, W., Meng, B., Lu, X., Liu, Y. and Tao, S. (2007) Extraction of
polycyclic aromatic hydrocarbons and organochlorine pesticides from soils: A comparison
between Soxhlet extraction, microwave-assisted extraction and accelerated
solvent extraction techniques. Analytica Chimica Acta. 602.
211–222.
Zhang, H.F., Yang, X.H. and Wang, Y. (2011) Microwave assisted
extraction of secondary metabolites from plants: Current status and future
directions. Trends in Food Science & Technology. 22, 672–688.
Received: June 11, 2020
Sent to Subject Editor: July 7, 2020
Accepted: January 3, 2021
Recommended by Subject Editor Mariano
Martín Martín

![]() ) was calculated
by Eq. 1, yield of eugenol (YE) was calculated by Eq. 2, and yield of global process (YP) by Eq. 3 for each of the method applied. The values were expressed
as mean±standard deviation (n=2).
) was calculated
by Eq. 1, yield of eugenol (YE) was calculated by Eq. 2, and yield of global process (YP) by Eq. 3 for each of the method applied. The values were expressed
as mean±standard deviation (n=2).![]() (1)
(1)![]() (2)
(2)![]() (3)
(3)